A) The melting point will decrease.
B) The melting point will remain the same.
C) The melting point will increase.
D) The substance will not melt at pressures of 1 atm and above; instead, the solid sublimes to form the gas phase.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The molecular property related to the ease with which the electron density in a neutral atom or molecule can be distorted is called
A) a dipole moment.
B) polarizability.
C) a dispersion force.
D) surface tension.
E) a van der Waals force.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
How much enthalpy is necessary to heat 10.0 g of solid benzene (C6H6)at 0.0°C to benzene vapor at 100°C? 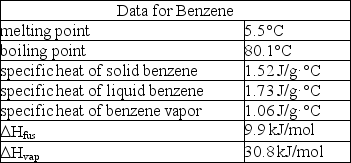
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following substances should have the lowest boiling point?
A) CBr4
B) CBr3F
C) CBr2F2
D) CBrF3
E) CF4
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
MgO has the same crystal structure as NaCl, face-centered cubic.How many oxide ions surround each Mg2+ ion as nearest neighbors?
A) 4
B) 6
C) 8
D) 10
E) 12
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Indicate all the types of intermolecular forces of attraction in C2H6(g).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of atoms in a body-centered cubic unit cell is
A) 1
B) 2
C) 3
D) 4
E) 8
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following liquids would have the highest viscosity at 25°C?
A) CH3OCH3
B) CH2Cl2
C) C2H5OH
D) CH3Br
E) HOCH2CH2OH
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the graph of vapor pressure to determine the normal boiling point of O2. 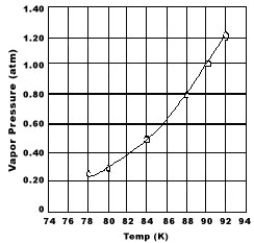
A) 92 K
B) 90 K
C) 88 K
D) 84 K
E) O2 doesn't boil because it is always a gas.
G) B) and E)
Correct Answer

verified
B
Correct Answer
verified
Short Answer
Indicate all the types of intermolecular forces of attraction in F2(l).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The zincblende structure of ZnS has the relatively large sulfide ions arranged at the lattice points of a face-centered cubic structure. The edge length of this cubic unit cell is 540.9 pm.Determine the density of zincblende.
A) 3.081 g/cm3
B) 1.023 g/cm3
C) 4.091 g/cm3
D) 2.046 g/cm3
E) 2.032 g/cm3
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Based on the phase diagram shown below, which is more dense: the liquid phase or the solid phase? 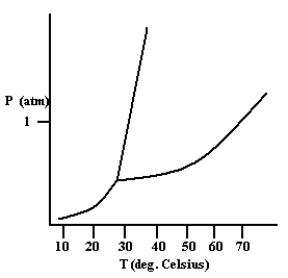
Correct Answer

verified
the solid ...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Given that the heat of vaporization of diethyl ether is 26.0 kJ/mol and the vapor pressure of diethyl ether is 440 torr at 20.°C, calculate the normal boiling point of diethyl ether.
Correct Answer

verified
36°C (the ...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
The vapor pressure of ethanol is 400 mmHg at 63.5°C. Its molar heat of vaporization is 39.3 kJ/mol. What is vapor pressure of ethanol, in mmHg, at 34.9°C?
A) 1,510 mmHg
B) 100 mmHg
C) 200 mmHg
D) 0.0099 mmHg
E) 4.61 mmHg
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Of the pair of compounds given, which would have the stronger intermolecular forces of attraction? HF or HCl
Correct Answer

verified
Correct Answer
verified
Short Answer
What phase exists at the point labeled a? 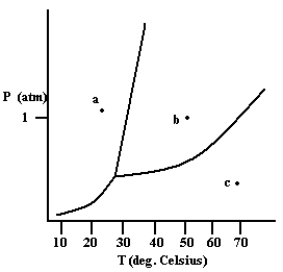
Correct Answer

verified
solid
Correct Answer
verified
Short Answer
Identify the dominant (strongest)type of intermolecular force present in NH3(l).
Correct Answer

verified
Correct Answer
verified
Short Answer
Given the following compound and its boiling point, identify whether it is polar or nonpolar: H2S, -60.7°C.
Correct Answer

verified
polar
Correct Answer
verified
Short Answer
Of the pair of compounds given, which would have the stronger intermolecular forces of attraction? H2S or H2Se
Correct Answer

verified
Correct Answer
verified
True/False
Ethanol (C2H5 - OH)will have a greater viscosity than ethylene glycol (HO - CH2CH2 - OH)at the same temperature.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 140
Related Exams