A) 3.87 m/s
B) 5990 m/s
C) 1.21 * 10-7 m/s
D) 3.26 m/s
E) 5.99 m/s
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the wavelength of a ball bearing with a mass of 10.0 g, and a velocity of 10.0 cm/s?
Correct Answer

verified
6.63 * 10<...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
A single pulse of a laser yields an average of 5.00 * 1018 photons with = 633 nm. If melting ice to water at 0°C requires 6.01 kJ/mol, what is the fewest number of laser pulses need to melt 10.0 g of ice?
A) 3830
B) 3340
C) 38300
D) 2120
E) 212
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons are there in the 3rd principal energy level (n = 3) of a phosphorus atom?
A) 3
B) 5.
C) 6
D) 8
E) 10
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the electron configuration of calcium?
Correct Answer

verified
1s22s22p63s23p...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
As the frequency of an electromagnetic wave increases
A) its speed must increase.
B) its wavelength must increase.
C) its amplitude must increase.
D) its energy must increase.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
The colors of the visible spectrum are blue, green, orange, red, violet, and yellow.Of these colors, _______ has the most energy.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A common way of initiating certain chemical reactions with light involves the generation of free halogen atoms in solution. If H for the reaction Cl2(g) 2Cl(g) is 242.8 kJ/mol, what is the longest wavelength of light that will produce free chlorine atoms in solution?
A) 246.3 nm
B) 465.2 nm
C) 349.3 nm
D) 698.6 nm
E) 492.6 nm
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is diamagnetic both in its ground state and in all of its excited states?
A) Mg
B) Ne
C) Cu
D) Zn
E) none of these
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A ground-state atom of iron has ___ unpaired electrons and is _____.
A) 0, diamagnetic
B) 6, diamagnetic
C) 3, paramagnetic
D) 5, paramagnetic
E) 4, paramagnetic
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the following diagram of a wave 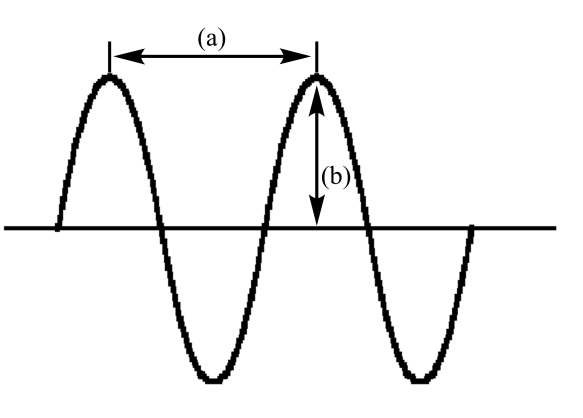
A) (a) is amplitude and (b) is wavelength
B) (a) is frequency and (b) is amplitude
C) (a) is wavelength and (b) is frequency
D) (a) is amplitude and (b) is frequency
E) (a) is wavelength and (b) is amplitude
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
An electron in a 3p orbital could have a value of 2 for its angular momentum quantum number (l).
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In an electron microscope, electrons are accelerated to great velocities. Calculate the wavelength of an electron traveling with a velocity of 7.0 * 103 kilometers per second. The mass of an electron is 9.1 * 10-28 g.
A) 1.0 * 10-13 m
B) 1.0 * 10-7 m
C) 1.0 m
D) 1.0 * 10-10 m
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following atoms is paramagnetic both in its ground state and in all of its excited states?
A) C
B) N
C) O
D) Ti
E) Cr
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A ground-state chromium atom has how many unpaired electrons?
A) 1
B) 2
C) 4
D) 5
E) 6
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A ground-state atom of arsenic has
A) no unpaired electrons.
B) one unpaired electron.
C) two unpaired electrons.
D) three unpaired electrons.
E) four unpaired electrons.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element has the following ground-state electron configuration? [Kr]5s24d105p3
A) Sn
B) Sb
C) Pb
D) Bi
E) Te
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element has the following ground-state electron configuration? [Kr]5s14d5
A) Mn
B) Mo
C) Nb
D) Re
E) Tc
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Write the ground state electron configuration for a lead atom.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the frequency of visible light having a wavelength of 486 nm.
A) 2.06 * 1014 /s
B) 2.06 * 106 /s
C) 6.17 * 1014 /s
D) 1.20 * 10-15 /s
E) 4.86 * 10-7 /s
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 115
Related Exams