A) A2I3
B) A3I2
C) AI3
D) A3I
E) AI
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the charge on the stable ion formed by selenium?
A) +2
B) +1
C) -1
D) -2
E) -3
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An element with the general electron configuration for its outermost electrons of ns2np1 would be in which element group?
A) 2A
B) 3A
C) 4A
D) 5A
E) 8A
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the element with the electron configuration [Xe]6s24f7. This element is
A) a representative element.
B) a lanthanide element.
C) a nonmetal.
D) an actinide element.
E) a noble gas.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Since zirconium is a metal, ZrO2 is expected to be a/an _____ oxide.
A) acidic
B) ionic
C) amphoteric
D) neutral
E) basic
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following ions in order of increasing ionic radius: K+, P3- , S2- , Cl- . increasing radius
A) K+ < Cl- < S2- < P3-
B) K+ < P3- < S2- < Cl-
C) P3- < S2- < Cl- < K+
D) Cl- < S2- < P3- < K+
E) Cl- < S2- < K+ < P3-
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the elements listed below has the following pattern for its first six ionization energies? (I1 = first ionization energy, I2 = second ionization energy, etc.) 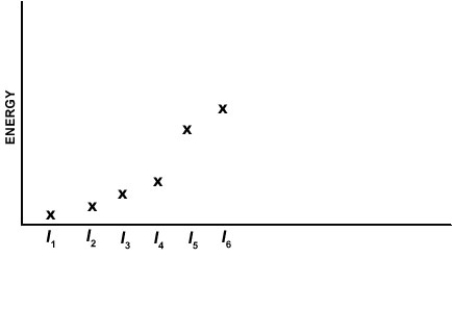
A) Ca
B) Si
C) Al
D) Se
E) P
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The general electron configuration for noble gas atoms is
A) ns2np6.
B) ns2np5.
C) ns2np4.
D) ns2np3.
E) ns2.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The chief contribution of physicist Henry Moseley to atomic theory was
A) the discovery of the periodic law.
B) the determination of the charge of the proton.
C) the measurement of the atomic numbers of the elements.
D) the scientist who determined the electric charge of the electron.
E) the discovery of the law of octaves.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements behaves chemically similarly to calcium?
A) magnesium
B) sodium
C) sulfur
D) chlorine
E) iron
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Which species has the greater radius, an I- ion or an I atom? Briefly explain your choice of answer.
Correct Answer

verified
I-; I and I- have the same number of proto...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
For which of the following reactions is the enthalpy change equal to the third ionization energy of vanadium?
A) V2+(g) V3+(g) + e-
B) V3+(g) + e- V2+(g)
C) V(g) V3+(g) + 3e-
D) V2-(g) + e- V3-(g)
E) V3+(g) V4+(g) + e-
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the electron configuration for the aluminum ion?
A) 1s22s22p63s2
B) 1s22s22p63s23p2
C) 1s22s22p63s23p1
D) 1s22s22p6
E) 1s22s22p63s23p4
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the charge on the monatomic ion that calcium forms in its compounds?
A) +2
B) +1
C) -1
D) -2
E) -3
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following pairs are isoelectronic?
A) Mn2+ and Ar
B) Zn2+ and Cu2+
C) Na+ and K+
D) Cl- and S
E) K+ and Cl-
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons are in the 4p orbitals of selenium?
A) 0
B) 2
C) 4
D) 5
E) 6
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For which of the following reactions is the enthalpy change equal to the second ionization energy of nitrogen?
A) N2+(g) N3+(g) + e-
B) N2+(g) + e- N+(g)
C) N(g) N2+(g) + 2e-
D) N-(g) + e- N2-(g)
E) N+(g) N2+(g) + e-
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements has the smallest ionization energy?
A) Li
B) Na
C) Be
D) K
E) Rb
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The elements in Group 2A are known by what name?
A) transition metals
B) halogens
C) alkali metals
D) alkaline earth metals
E) noble gases
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an amphoteric oxide?
A) Na2O
B) MgO
C) Al2O3
D) SO2
E) Cl2O7
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 119
Related Exams