A) 1
B) 2
C) 3
D) 4
E) 5
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The proton(s) on which C will produce a splitting pattern that is a doublet in its 1H NMR spectrum? 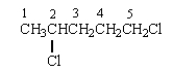
A) 1
B) 2
C) 3
D) 4
E) 5
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules has more than three (3) different types of protons in its 1H NMR? 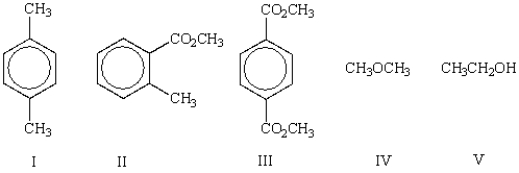
A) I
B) II
C) III
D) IV
E) V
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is most likely to absorb ultraviolet radiation in the range of 200 to 400 nm?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An IR spectrum of an unknown organic molecule having the molecular formula of C4H8O shows no absorptions at 1630-1780 cm-1 or at 3200-3500 cm-1.To what class of compounds does the unknown belong?
A) alcohol
B) carboxylic acid
C) ketone
D) ether
E) aldehyde
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A hydrocarbon shows a parent ion peak in its mass spectrum at m/z 102.Its 1H NMR spectrum shows only two peaks, a singlet at 2.7 (1H) and a multiplet at 7.4 (5H) .The correct structure is:
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Bending vibrations in the infared region occur at:
A) 3000 cm-1
B) 2200 cm-1
C) 1700 cm-1
D) below 1400 cm-1
E) over 3000 cm-1
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A compound C3H3Cl5 has a 1H NMR spectrum that consists of a triplet at 4.5 and a doublet at 6.0 (J = 7 Hz) with relative areas 1:2.Its structure is
A) CH3CCl2CCl3
B) CH2ClCCl2CHCl2
C) CHCl2CCl2CCH2Cl
D) CHCl2CHClCHCl2
E) CH2ClCHClCCl3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following compound will have an 1H NMR spectrum that consists of: 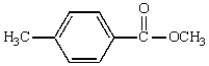
A) two singlets, area ratio 6:4.
B) three singlets, area ratio 3:3:4.
C) four singlets, area ratio 3:3:2:2.
D) two singlets and two doublets, area ratio 3:3:2:2.
E) two triplets and a singlet, area ratio 3:3:4.
G) D) and E)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Which of following will have three peaks in its 1H decoupled 13C NMR spectrum?
A) pentane
B) isopentane
C) isobutane
D) benzene
E) m-chlorotoluene
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds will have one peak in its 1H NMR and two peaks in its 13C NMR spectrum?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following compound will have a proton NMR spectrum that shows: 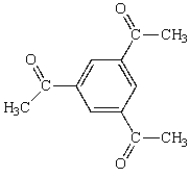
A) two singlets, area ratio 3:1.
B) two singlets, area ratio 9:1.
C) three singlets, area ratio 3:3:6.
D) four singlets, area ratio 3:2:6:1.
E) six singlets, area ratio 3:3:3:1:1:1.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which bond would show a strong, broad absorption in the IR between 3600 and 3200 cm-1?
A) O-H
B) C-Cl
C) C=O
D) C-H
E) C-O
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Compounds that will show only two sharp singlets in their 1H NMR spectra are: 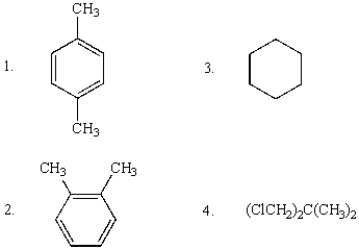
A) 1 and 2
B) 3
C) 1 and 3
D) 1 and 4
E) 1, 2, and 4
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules will show two peaks with an area ratio of 6:4 in its 1H NMR spectrum? 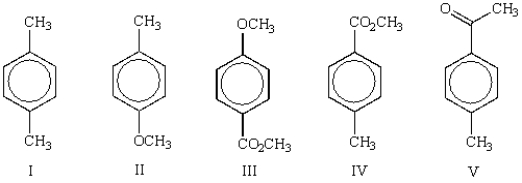
A) I
B) II
C) III
D) IV
E) V
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the 1H NMR spectrum of the following molecule, the protons on which carbon will have the most downfield chemical shift? 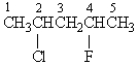
A) 1
B) 2
C) 3
D) 4
E) 5
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
As the wavelength of the radiation decreases, the increases.
A) energy
B) frequency
C) wave number
D) all of these
E) all decrease
G) A) and B)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Which of the following molecules would absorb at the longest wavelength in the ultraviolet region?
A) hexane
B) 2-hexene
C) 2,4-hexadiene
D) 1,3,5-hexatriene
E) cyclohexane
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which molecule will produce two lines in the proton decoupled 13C NMR spectrum?
A) CH3CH2OH
B) CH3OCH3
C) CH3CH2CH2OH
D) CH3OCH2CH3
E) ![]()
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The area in the infared spectrum between 1400 and 400 cm-1 is called the .
A) solid region
B) fingerprint region
C) functional group region
D) location region
E) none of these
G) B) and D)
Correct Answer

verified
B
Correct Answer
verified
Showing 1 - 20 of 48
Related Exams